Abstract
Background For the majority of patients with low risk myelodysplastic syndromes (MDS), anaemia is the most significant clinical consequence. Currently there are very limited options beyond erythropoiesis stimulating agents (ESAs) and transfusions for these patients. Luspatercept is a first in class erythroid maturation agent which binds to transforming growth factor β (TGF β ) superfamily ligands, approved for use in patients with low risk MDS in the EU and USA, who require RBC transfusions.
Aim We aimed to uncover the underlying dysregulated pathways in erythroid development in low risk MDS, in order to identify predictors of Luspatercept response and aid the development of new therapeutic strategies.
Methods We performed single cell (sc) RNA sequencing using the 10x genomics platform in 5 patients with low risk MDS related anaemia and a normal karyotype. Mutation profiles varied between patients and included mutations in splice factor genes. In one patient, we analysed serial samples before and after Luspatercept therapy; at the time of transfusion independence and then at loss of response. We compared to 5 healthy control (HC) samples analysing both live mononuclear cells and CD34 enriched viable bone marrow cells from our patients and the HCs.
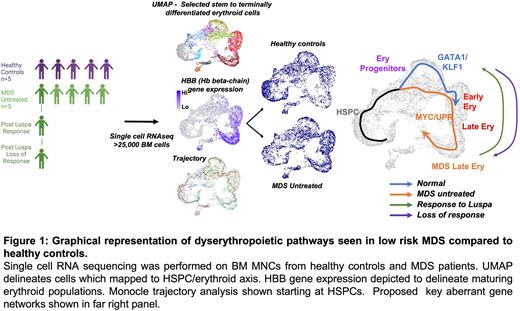
Results Following unsupervised clustering of all 27,610 cells, we focused all of our subsequent analysis on cell populations we identified as belonging to the stem-erythroid developmental trajectory, based on established marker gene expression. The most striking distinction between MDS and HC cells, was the presence of distinct subpopulations of maturing erythroid cells, which were significantly enriched in the untreated MDS patients (OR range 4.4 - 12.5, p < 10e-64). In order to better understand how the development of erythropoiesis differs in MDS compared to normal, we performed trajectory analysis which revealed that MDS cells preferentially undertook a distinct route of erythropoiesis . Furthermore, we pinpointed the divergence in MDS haematopoiesis from normal, which occurred in erythroid progenitors (EPs). Whereas normal erythropoiesis engaged a GATA1/KLF1 regulon axis (OR: 3.94, p < 10e-76), MDS EPs inefficiently upregulated GATA1/KLF1 regulons (OR: 1.45, p = 0.007) and upregulated MYC replicative stress response and unfolded protein response (UPR) gene networks (fold-change: 1.52, p < 10e-16).
Focussing on the EP populations enriched in the MDS cells, we observed upregulation of iron handling and amino acid metabolism gene networks. These networks remain aberrantly highly expressed in MDS terminally differentiated erythroid cells, whereas in the normal setting, these networks are downregulated prior to these late developmental stages. In addition, our data suggests that the most terminally differentiated of the late erythroid MDS cells upregulate telomerase maintenance and endoplasmic reticulum (ER) stress response pathways. We then investigated the impact of Luspatercept treatment on the regulation of these pathways at the time of transfusion independence. At the time of response, Luspatercept downregulated the MYC replicative stress response and UPR pathways aberrantly upregulated in MDS (fold-change: 0.84, p = 6e-07) and decreased the size of the most terminally differentiated dyserythropoietic populations (OR range: 0.25 - 0.4, p < 10e-5), prominent in the untreated MDS samples, restoring a pattern more similar to normal erythropoiesis. Of note, at loss of response to Luspatercept, these changes reverted towards levels expressed in the pre-treatment MDS sample, advocating the importance of these populations and pathways as biomarkers of response.
Conclusion Single cell RNA sequencing revealed an altered erythroid differentiation landscape in patients with low risk myelodysplastic syndromes, delineating new dyserythropoietic subpopulations, driven by the dysregulation of key molecular pathways (Figure 1). Our data indicates that Luspatercept promotes the restoration of normal erythropoiesis by downregulating key pathways beyond the TGF beta pathway.
Disclosures
Psaila:Constellation Therapeutics: Consultancy; Alethiomics: Consultancy, Current equity holder in private company, Honoraria, Other: Co-founder , Research Funding; Blueprint Therapeutics: Consultancy; Novartis: Consultancy, Honoraria, Speakers Bureau; Galecto: Research Funding; Evotec: Research Funding. Mead:Incyte: Consultancy, Speakers Bureau; CTI: Consultancy, Speakers Bureau; Sierra Oncology: Consultancy, Speakers Bureau; Gilead: Consultancy, Speakers Bureau; Sensyn: Consultancy, Speakers Bureau; Galecto: Consultancy, Research Funding, Speakers Bureau; Pfizer: Consultancy, Speakers Bureau; Alethiomics Ltd: Consultancy, Current equity holder in private company, Other: Co-founder and equity holder, Research Funding; Karyopharm: Consultancy, Speakers Bureau; AbbVie: Consultancy, Speakers Bureau; Celgene/BMS: Consultancy, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Research Funding, Speakers Bureau. Quek:Servier: Research Funding; Bristol Myers Squibb: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal